Preliminary 18-month safety and effectiveness results of RNS System responsive thalamic stimulation for the treatment of idiopathic generalized epilepsy (IGE) poster at AES 202511
CAUTION Investigational device. Limited by U.S. law to investigational use for Idiopathic Generalized Epilepsy.
 Download Poster
Download Poster
Conclusive Evidence.
Long-Term Seizure
Reduction
82% median seizure reduction at 3 years1
Rapid Seizure
Reduction
62% median seizure reduction at 6 months1
Results from the RNS System Post-Approval Study, the largest FDA-reviewed prospective neuromodulation study in focal drug-resistant epilepsy, confirm rapid and sustained seizure control, with outcomes from 324 implanted patients at 32 centers across the U.S.1,6,7
Seizure Freedom is Possible
42% of patients seizure free for 6+ months1,9
(n=268)
Seizure Freedom at Last Follow-up
Stay at the forefront of epilepsy treatment.
Sign up for the NeuroPace newsletter to receive updates on the latest research and innovations.
Note: Therapies were studied using different study designs. Caution must be exercised when comparing results.
- RNS System Post-approval Study Oral Presentation, American Academy of Neurology, April 2025, all outcomes are ITT, median seizure reduction is observed case data, seizure freedom at last follow-up is LOCF
- Morris et al, Neurology, 1999
- Kaufmann et al., Epilepsia, 2024
- VNS Prospective Trial (E01, E04 Pooled): https://www.accessdata.fda.gov/cdrh_docs/pdf/p970003.pdf
- Peltola et al., Neurology, 2023
- Salanova et al., 2015
- DeGiorgio et al, Epilepsia, 2000
- Englot et al, Neurosurgery, 2016
- At some point during the study
- Compared to placebo arm of ASM trial. 2.3/1000 patient years rate of SUDEP vs 6.9/1000 patient years rate of SUDEP in Anti-seizure Medication (ASM) placebo; Ryvlin et al, Lancet Neurol, 2011
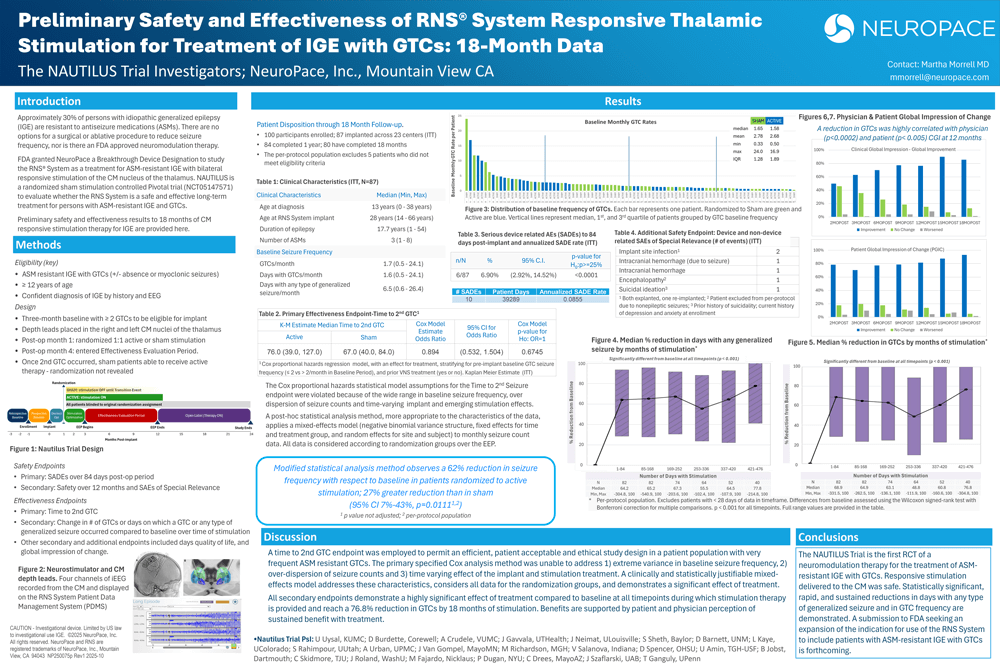
- Preliminary Safety and Effectiveness of RNS System Responsive Thalamic Stimulation for Treatment of IGE with GTCs: 18-Month Data, American Epilepsy Society, Dec 2025.
Rx Only. The RNS® System is an adjunctive therapy for adults with refractory, partial onset seizures with no more than 2 epileptogenic foci. See important safety information at https://www.neuropace.com/safety/. Refer to the labeling for a description of the RNS® System and its components, indications for use, contraindications, warnings, cautions, adverse events and instructions for use. ©2025 NeuroPace, Inc. All rights reserved. NeuroPace and RNS are registered trademarks of NeuroPace, Inc., Mountain View, CA 94043. NP 200156 Rev 2 / Rev. Date: 2025-03